Two well-studied autism mutations cause social defects in mice. But they’ve now been found to do so through effects outside the brain. How can this be?

“Autism is about having a pure heart and being very sensitive… It is about finding a way to survive in an overwhelming, confusing world… It is about developing differently, in a different pace and with different leaps,” Trisha Van Berkel.
How do genes—DNA, these physical atoms: carbons, hydrogens, and nitrogens—influence our subjective experience of consciousness? And how can mutations to genes lead to the alterations in behavior and consciousness seen in disorders like autism?
Autism Spectrum Disorder (ASD) is now estimated to affect 1 in 68 children in the United States and causes difficulties with social interaction, and a tendency towards repetitive behaviors and restricted interests. A surprising new study published in Cell indicates that two well-studied mutations that cause autism may actually be acting outside of the brain, and an oft-overlooked symptom may be more important than we’ve thought.
This new study—which we’ll return to in a moment—connects fundamental autism biological research to an emerging line of research that shows our body contains a special set of nerves whose function is detecting pleasant social touch.
Orphaned monkeys, cattle squeeze chutes, and hypersensitivity to touch.
Touch is an integral part of social experience. In the 1950s, In response to radical behaviorists like B.F. Skinner, who thought that a child’s love for his or her mother came from the repeated association of the mother with food, Harlow showed that monkeys have an intrinsic desire for touch:
Those poor cute Harlow monkeys… 😦
Harlow raised baby monkeys in complete social isolation, unless you count the two monkey mannequins in their room. The first was made of metal wire and dispensed milk, and the second had no clear functional benefit to the baby monkey, but it was covered in soft cloth the monkey could cling to. Harlow showed the monkeys would feed from the metal monkey, but spend their time with the cloth one and seek it out when they were distressed.
Parental touch plays an important role in soothing a child’s negative emotions[1], and is a large component of early interactions between parent and child such as nursing, cradling, and swaddling, the foundation upon which social interaction may begin to be learned.
However, many autistic people report difficulties with their sense of touch. Temple Grandin, a neuroscientist on the spectrum described her experience: “As a child, I craved to feel the comfort of being held, but I would pull away when people hugged me. When hugged, an overwhelming tidal wave of sensation flowed through me.” This desire to be embraced (and some bovine inspiration) prompted her to invent a “squeeze machine,” that allowed her to simulate being embraced without the overwhelming, unpredictable stimulation of direct human contact.
While Grandin isn’t representative of the typical ASD patient, her experiences with touch are. In a survey of caregivers of nearly 300 children with ASD, 60% reported their child experienced tactile sensitivity differences greater than two standard deviations from the mean—for example experience severe distress during grooming. These symptoms are now become acknowledged as representative, and the latest version of Psychiatry’s Diagnostic and Statistic Manual (DSM) includes sensory abnormalities as a core feature of the disorder.
 source: https://doi.org/10.5014/ajot.61.2.190
source: https://doi.org/10.5014/ajot.61.2.190
So, what is causing these sensory differences in autism? Why is touch important for human interaction and social development? Can social deficits in autism be explained by sensory hypersensitivity?
What makes touch feel pleasant? – the Recently-Described Affective Touch System
What makes the gentle touch of fingers sensual, a hug comforting? Researchers have recently discovered an affective touch system, specialized for detecting pleasant touch.
This system begins all the way out in the skin, with a special set of sensory nerves tuned to detect social touch at a cellular level, because they respond maximally to a set of stimuli that matches pleasant touch. These nerves, known as C tactile (CT) afferents, integrate temperature and pressure information, respond maximally to skin temperature stimulation, light intensity touch, and the stroking speeds that feel the most pleasant and soothing[2]. These neurons also communicate too slowly to the brain to be useful for the conventional tactile tasks such as discriminating shape or texture.
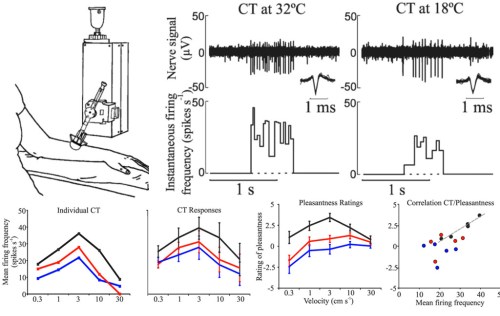
Yes, scientists made a robot to stroke people’s skin because… science. And because reliable, replicable stimuli are good, mmkay? Modified from Ackerly et al. 2014.
Okay, so CT afferents seem to be nerves specialized for detecting social touch, but how is that information processed? Thanks to an interesting patient, scientists could also investigate where in the brain these affective touch signals are transmitted. Due to a rare disease, the patient lost her normal discriminatory tactile nerves, but maintained her affective touch CT afferent nerves. Her unique condition meant that when scientists rubbed her arm, they exclusively stimulated the CT afferents, without stimulating other tactile sensory neurons.
So, as cognitive neuroscientists are wont to do, they stuck her in an fMRI and rubbed her arm to see what brain areas were processing affective touch signals. Interestingly, stimulating CT fibers did not active the somatosensory cortex, which is normally activated by touch and is important in sensory discrimination. Instead, the stimulation of CT fibers led to activation of the insular cortex, a brain area involved in many functions, including emotional regulation and social processing. Earlier this year, this system was observed through brain recordings of insular neurons in monkeys, indicating it is an evolutionary conserved and therefore important system. The differential processing of CT fiber touch in the brain further supports the idea these nerves are part of a specialized for detecting pleasant social touch. This pleasent touch system may help explain the many effects massage, which include decreasing the stress hormone cortisol and modifying levels of neuromodulators like serotonin and dopamine.
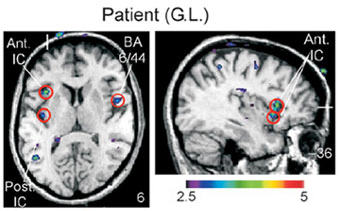
Modified from Oloussan et al., 2002
What is causes tactile sensory differences these differences in autism?
ASD Mouse Models
Autism Spectrum Disorders represent an incredibly heterogeneous group of disorders that as of 2015 experts estimated are influenced by 200-1000 genes (that’s up to 5% of all protein-coding genes). ASD doesn’t appear to be entirely genetic–studies of identical twins show that when one twin is diagnosed, the other has somewhere between a 30-90% chance of suffering autism as well[3].
In rare cases of autism, mutations to single genes or the loss small groups of neighboring genes greatly increase the risk of autism spectrum disorders, such as Rett syndrome, Fragile X, or Phelan-McDermid syndrome, to the extent that this mutations are often considered causative. These ASD syndromes are also often associated with other congenital conditions including intellectual disability.
These same mutations, or equivalent mutations, have been recreated in mice creating ASD mouse models that show modified physiology and behavior, including social behavior deficits—for example these mice may consistently ignore other mice. Translating biology from mice back to humans in always tricky, let alone trying to understand the complex process of social interaction, but the social deficits seen in autistic model mice give hope that these models may help understand ASD.
New Study on Hypersensitivity to touch in ASD Mouse Models
The new study published in Cell, titled Peripheral Mechanosensory Neuron Dysfunction Underlies Behavioral Deficits in Mouse Models of ASDs (a mouthful, right?), shows that mice modeling the aforementioned syndromes—Rett, Fragile X, Phelan-McDermid—and mice bearing a mutation associated with autism that affects inhibitory GABA receptor subunit B3 (GABRB3)—all show tactile hypersensitivity and tactile sensory impairment.
The researchers then investigated the mechanisms of this impairment in two of the mouse models, the Rett Syndrome model mice which have a mutation to MECP2 and the GABRB3 mutants. They showed that when the mutations are induced only in the cortex, the outermost layer of the brain thought to be involved in higher cognition, the tactile deficits were not observed. However, when they induced the mutations exclusively in somatosensory neurons, which include tactile neurons, they saw tactile hypersensitivity in the mice[4]! So the tactile-hypersensitivity phenotype of these mice is coming from outside the brain[5]! So sure, peripheral nerve effects change the sense of touch, but can explain the anxiety and social dysfunctions seen in these mice as well? Yes.
In a spectacular set of experiments, the investigators first showed that deleting gene, whose loss causes Rett-syndrome, MECP2, only in peripheral nerves[6] creates the same social deficits and anxiety behaviors seen when the gene is deleted from the whole mouse. Then in the knockout mice, where the gene had been deleted from every tissue, they showed restoring it in just the peripheral nerves was sufficient to “rescue” the mouse, and prevent the aberrant social and anxiety behaviors!
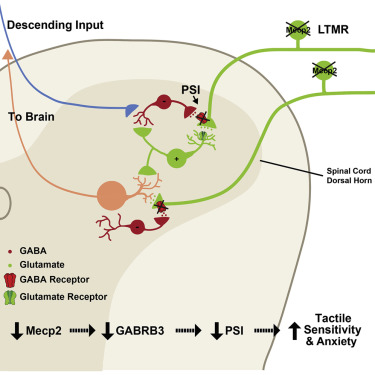 Source: Orefice et al., 2016
Source: Orefice et al., 2016
But let’s hold up a second before we get too excited. One way of interpreting these experiments is that these mutations caused tactile sensory problems which led to social deficits and when they sensory problems were fixed the social deficits were fixed as well. An alternate explanation, however, is that maybe the mice never had social deficits in the first place (or had more subtle ones not measurable by the behavioral test used), but just tactile impairments that were being misinterpreted as social deficits—kind of akin to asking a blind person to distinguish between to pictures and calling him stupid when he can’t[7]. (Though again, as discussed above this same tactile impairment may contribute to the social phenotypes seen in humans with ASD.)
I am also curious to see if sensory hypersensitivity can explain the repetitive grooming behaviors observed in models of ASD like Shank3B mutant mice. These mice will groom themselves until they lose their hair and develop injuries, which has been compared to the repetitive behaviors of autism or even compulsive behaviors of OCD, but maybe just because it is occurring at the animals, one could imagine peripheral nerve involvement.
As an aside, just like we shouldn’t read too much into “behavioral deficits” observed in model mice, we shouldn’t pathologize unusual behavior in humans, just because we can’t understand it. The following video by AM Baggs, an artist and activist with ASD, should be required viewing for anyone with an interest in autism:
(For those pressed for time, the artist’s explanation begins at 3:12, but I recommend watching the whole thing)
Can social deficits in autism be explained by sensory hypersensitivity?
We know from studies of orphanages in Eastern Europe, where historically children experienced little touch and other human interaction, that these children went on to experience deficits in cognitive function, problems with language, and even what some have termed quasi-autism or institutional autism. Of course these children were deprived of much more than social touch—they were also deprived of attention, human interaction, and all but the most basic of care.
However, one could imagine that for a child with ASD, who experiences hypersensitivity to touch, that the social touch normally beneficial during infancy could be masked, distorted, or even turned harmful by this sensory hypersensitivity. These unpleasant experiences could lead to avoidance of touch and social interaction, and it’s easy to imagine, as psychodynamic (Freudian) therapists do, that strong experiences in early childhood could have compounding and profound effects throughout life.
I’m not trying to say I think there’s any evidence to argue that sensory hypersensitivity causes ASD. ASDs again are complex disorders and the genes and processes involved probably affect a range of tissues throughout the brain, and—as we saw here—outside the brain as well. But the effect of tactile hypersensitivity, and how it may impair children receiving and benefiting from social touch is something we need to think about and further research.
Conclusions
I think the first take home message of this story is that we shouldn’t ignore things going on outside the brain, or at “low-levels” of the brain, and focus all our attention on the areas that supposedly mediate “higher” cognition. The embodiment of the brain, and inputs the brain receives are extremely important for proper brain development and function. We are not brains floating in jars à la Futurama, but rather the loop between our brains, bodies, and environment.
To me the second take home message of this study and affective touch research is something that people like Temple Grandin have known intuitively for years–that therapeutic touch, or other research into the tactile hypersensitivity, might be a useful treatment for ASD. Scientists are already looking into this issue. A 2011 meta-analysis of message therapy for children with ASD concluded: “Limited evidence exists for the effectiveness of massage as a symptomatic treatment of autism. Because the risk of bias was high, firm conclusions cannot be drawn. Future, more rigorous randomized clinical trials seem to be warranted.” Perhaps, if we better understand the biological basis behind these hypersensitivities and the benefits of affective touch we can tailor more effective therapeutic touch therapies, or design drugs to treat tactile hypersensitivity.
But maybe we just need to take a step back from our own assumptions about how we think things feel and ask individuals with ASD, “What feels good to you?”
So what do you think?
Do you or does someone you know experience hyper-sensitivity to touch? How would you describe it? And does it make social touch difficult? Remove the desire for that kind of feeling?
Do you think that these genes are having their main effect inside or outside the brain. It does seem a strange coincidence for all the genes to have this same touch-hypersensitivity effect, and then also just happen to cause autism in people.
Though perhaps this is just a peripheral place that is acutely sensitive to excitation/inhibition imbalance, and similar but hard-to-test changes are occur in mice and human brains, and those are more responsible for autism’s core symptoms?
If you liked this article, please share it, and follow me to see more like it. I do this for free as a passion project and because of my insatiable need for validation from strangers over the internet. If you didn’t like it, tell me why, I’m also an insatiable glutton for punishment.
Footnotes (Asides)
[1] A review of these concepts is available in Hertenstein et al. 2006, http://www.ncbi.nlm.nih.gov/pubmed/17345871
[2]In contrast, other tactile sensory neurons increase their activity in response to speed, show no temperature modulation, and relay their information to the brain more quickly, consistent with their role in tactile discrimination.
[3] Keep in mind that just because it isn’t entirely genetic, doesn’t mean it can be controlled by child-rearing—non-genetic biological factors may play important roles
[4] To my understanding, the tests used in this study looked at effects on the tactile fibers and not the affect fibers specifically. It’s unclear if these mutations affect the social CT-afferent fibers as well.
[5] The investigators further examined the role of GABRB3 in Rett mutant mice, and showed that GABRB3 levels were decreased in tactile afferents in the spinal cord. They measured the electrical activity of the neurons these tactile afferents communicate to in the spinal cord and showed they are overactive, suggesting that the decrease in inhibitory GABA signaling causes overexcitement of the tactile nerve. This fit’s with the excitation-inhibition imbalance theory of autism, though interestingly in this case the imbalance is taking place outside the brain!
[6] In an earlier version of this blog post I mistakenly reported that these mice showed off-target CRE expression in the VTA, however I was confusing which Advillin-CRE line was being used. With the line used in this study, authors reported they didn’t see expression in the brain and excluded any animals that showed signs of ‘off-target’ CRE expression.
[7] Scientists may have made similar mistakes with Alz. model mice, testing the mice’s visual memory with stimuli which the mice could not distinguish due to their strain’s retinal degeneration and conflating vision loss for memory loss.
Filed under: Uncategorized | Leave a Comment

No Responses Yet to “Two well-studied autism mutations cause social defects in mice. But they’ve now been found to do so through effects outside the brain. How can this be?”